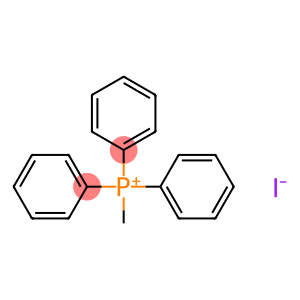
(Methyl)triphenylphosphonium Iodide-d3,13CD3
(Methyl)triphenylphosphonium Iodide-d3,13CD3
CAS: 282107-30-6
Molecular Formula: C19H18IP
(Methyl)triphenylphosphonium Iodide-d3,13CD3 - Names and Identifiers
| Name | (Methyl)triphenylphosphonium Iodide-d3,13CD3 |
| Synonyms | (Methyl)triphenyl- (Methyl)triphenylphosphonium Iodide-d3,13CD3 [2H3,13C,2H3]- (Methyl)triphenylphosphonium Iodide |
| CAS | 282107-30-6 |
(Methyl)triphenylphosphonium Iodide-d3,13CD3 - Physico-chemical Properties
| Molecular Formula | C19H18IP |
| Molar Mass | 404.224451 |
| Melting Point | >182°C (dec.) |
| Solubility | Chloroform (Slightly), Methanol (Slightly, Sonicated) |
| Appearance | Solid |
| Color | White |
| Storage Condition | Hygroscopic, -20°C Freezer, Under inert atmosphere |
(Methyl)triphenylphosphonium Iodide-d3,13CD3 - Introduction
(Methyl)triphenylphosphonium Iodide-d3,13CD3 is a salt containing iodide and a (methyl)triphenylphosphonium ion loaded with deuterium, 13C (D-deuterium, 13C-carbon). The following is a detailed description of its nature, use, preparation and safety information:
Nature:
-Appearance: White to light yellow solid
-Molecular formula: C19H16I13CD3P
-Molecular weight: 852.07g/mol
-Melting Point: 144-150 ° C
-Insoluble in water, soluble in organic solvents such as ether or chloroform
-Stable in the air and can be stored for a long time
Use:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 can be used for nuclear magnetic resonance (NMR) studies. The deuterium and 13C isotopic labels therein provide important information on the structure and reaction mechanism of the compounds.
-It can be used as a standard reagent to verify the NMR spectrum of other impurities.
Preparation Method:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 is usually synthesized by chemical synthesis. Specific preparation steps include:
1. Firstly, (methyl) triphenylphosphate reacts with methyl bromide or methyl iodide to generate (methyl)triphenylphosphonium bromide.
2. Next, (methyl)triphenyl phosphonium bromide is reacted with synthetic (deuterated, 13C-labeled) iodide to give (Methyl)triphenyl phosphonium Iodide-d3,13CD3.
Safety Information:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 is a chemical, and necessary safety procedures should be followed when using it.
-It may be irritating and damaging to the skin, eyes, respiratory tract and digestive system and should be avoided.
-Wear appropriate personal protective equipment such as goggles, protective gloves and lab coats during use.
-Store it in a dry, well-ventilated place, away from fire and oxidizing agents.
Nature:
-Appearance: White to light yellow solid
-Molecular formula: C19H16I13CD3P
-Molecular weight: 852.07g/mol
-Melting Point: 144-150 ° C
-Insoluble in water, soluble in organic solvents such as ether or chloroform
-Stable in the air and can be stored for a long time
Use:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 can be used for nuclear magnetic resonance (NMR) studies. The deuterium and 13C isotopic labels therein provide important information on the structure and reaction mechanism of the compounds.
-It can be used as a standard reagent to verify the NMR spectrum of other impurities.
Preparation Method:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 is usually synthesized by chemical synthesis. Specific preparation steps include:
1. Firstly, (methyl) triphenylphosphate reacts with methyl bromide or methyl iodide to generate (methyl)triphenylphosphonium bromide.
2. Next, (methyl)triphenyl phosphonium bromide is reacted with synthetic (deuterated, 13C-labeled) iodide to give (Methyl)triphenyl phosphonium Iodide-d3,13CD3.
Safety Information:
- (Methyl)triphenylphosphonium Iodide-d3,13CD3 is a chemical, and necessary safety procedures should be followed when using it.
-It may be irritating and damaging to the skin, eyes, respiratory tract and digestive system and should be avoided.
-Wear appropriate personal protective equipment such as goggles, protective gloves and lab coats during use.
-Store it in a dry, well-ventilated place, away from fire and oxidizing agents.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: methyl(triphenyl)phosphonium iodide Request for quotation
CAS: 282107-30-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 282107-30-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: (Methyl)triphenylphosphonium Iodide-d3,13CD3 Visit Supplier Webpage Request for quotation
CAS: 282107-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 282107-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: methyl(triphenyl)phosphonium iodide Request for quotation
CAS: 282107-30-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 282107-30-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: (Methyl)triphenylphosphonium Iodide-d3,13CD3 Visit Supplier Webpage Request for quotation
CAS: 282107-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 282107-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

